 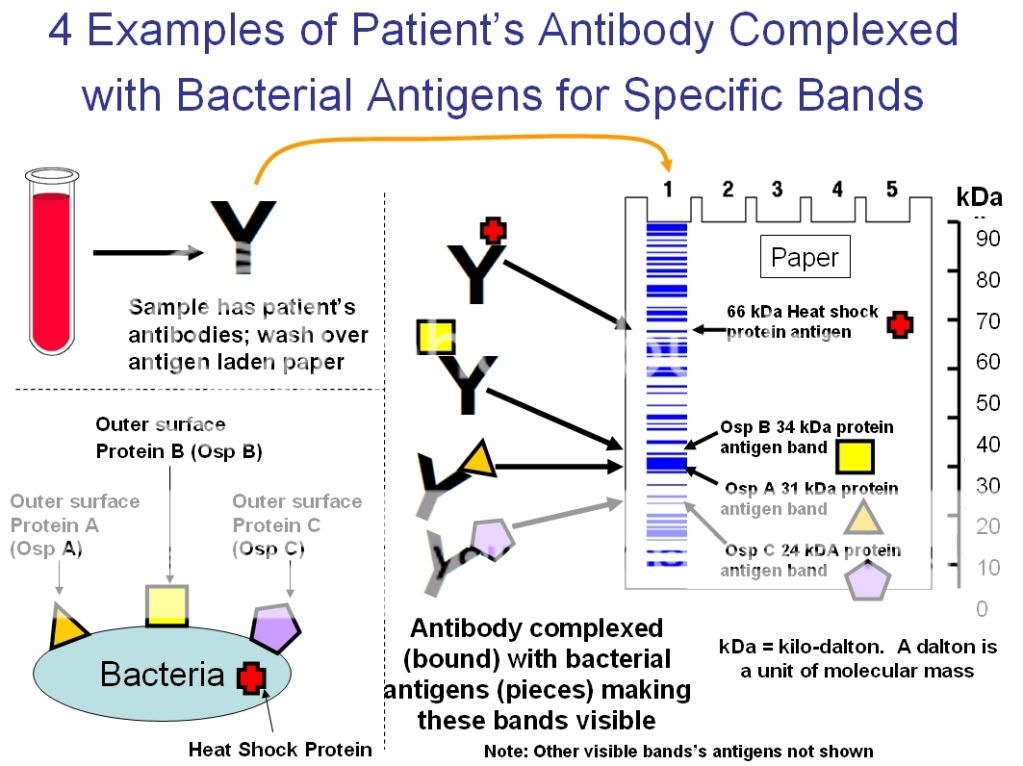
These tests are not approved by FDA because they have not been proven to be accurate. Some laboratories offer Lyme disease testing using urine or other body fluids. Such results may lead to people being treated for Lyme disease when they do not have it, instead of being treated for the true cause of their illness. Using the Western blot on its own makes a false positive result more likely. If the Western blot is negative, it suggests that the ELISA test was a false positive.ĬDC does not recommend testing by Western blot without first using the ELISA. Used appropriately, the Western blot test is designed to be “specific,” meaning that it will usually be positive only if a person has been truly infected by B. If the screening test is positive or inconclusive, a Western blot test should be performed to confirm the results. If the screening test is negative, it is highly unlikely that the person has Lyme disease and no further testing is recommended. The ELISA is designed to be very “sensitive,” meaning that almost everyone who has Lyme disease (and some people who do not) will test positive. A positive result from this first-level screening may suggest current or past infection. The ELISA is a blood test that detects antibodies but does not test for B. The Centers for Disease Control and Prevention (CDC) currently recommends a two-tiered testing approach: a conventional enzyme-linked immunoassay (ELISA) test, followed by a Western Blot test. To diagnose Lyme disease, a healthcare provider usually uses laboratory tests approved by the Food and Drug Administration (FDA) combined with information about a patient’s symptoms. These issues highlight the need for diagnostic tools that can distinguish between Lyme and other bacteria, and can find evidence of Lyme disease soon after infection. Unfortunately, other bacterial infections may mistakenly be reported as a positive Lyme antibody test. This gap between being infected with the bacteria and the body's creation of antibodies can result in a false negative test for those with very early Lyme disease, resulting in a delay in treatment.Ĭonversely, it is possible to receive a false positive test when a person does not actually have Lyme disease. But it can take a few weeks before a person's immune system creates measurable levels of antibodies. Lyme antibody tests-the most common diagnostic tool available today-look for antibodies in the blood that show a person has been exposed to B. Current diagnostic tests have some limitations that make a clear, quick diagnosis difficult. Because treatment is generally more effective in early stages of the disease, it is important to develop new tools that can help doctors make an early diagnosis.ĭoctors need to know whether a patient has an active infection, or has been exposed to the Lyme bacteria earlier in their life. Also, current diagnostic tests do not always detect early Lyme disease since antibodies take time to rise to detectable levels. The most distinct symptom of Lyme disease-the circular red rash known as erythema migrans (EM)-does not appear in at least one quarter of people who are actually infected with Lyme bacteria. Many of the common symptoms associated with the disease, such as headaches, dizziness, and joint/body pain, also occur with other diseases. Lyme disease can be difficult to diagnose for a number of reasons. Priorities include finding potential targets-substances that new diagnostic tools might measure in patient samples-and improving the sensitivity and specificity of currently available diagnostic tests, thereby leading to more accurate results. NIAID is committed to improving Lyme disease diagnostics by supporting innovative research projects. There is a great need to develop rapid, point-of-care tests to determine whether people are infected with Borrelia burgdorferi, the bacterium that causes Lyme disease. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
